 
Elements that exhibit similar chemistry appear in vertical columns called groups (numbered 1–18 from left to right) the seven horizontal rows are called periods. The periodic table is an arrangement of the elements in order of increasing atomic number. As expected, semimetals exhibit properties intermediate between metals and nonmetals. Most solid nonmetals are brittle, so they break into small pieces when hit with a hammer or pulled into a wire. Nonmetals can be gases (such as chlorine), liquids (such as bromine), or solids (such as iodine) at room temperature and pressure. Nonmetals, in contrast, are generally poor conductors of heat and electricity and are not lustrous. Of the metals, only mercury is a liquid at room temperature and pressure all the rest are solids. The vast majority of the known elements are metals. Metals-such as copper or gold-are good conductors of electricity and heat they can be pulled into wires because they are ductile they can be hammered or pressed into thin sheets or foils because they are malleable and most have a shiny appearance, so they are lustrous. The distinction between metals and nonmetals is one of the most fundamental in chemistry. As you might expect, elements colored in gold that lie along the diagonal line exhibit properties intermediate between metals and nonmetals they are called semimetals. The heavy orange zigzag line running diagonally from the upper left to the lower right through groups 13–16 in divides the elements into metals (in blue, below and to the left of the line) and nonmetals (in bronze, above and to the right of the line). The semimetals lie along a diagonal line separating the metals and nonmetals. The metals are on the bottom left in the periodic table, and the nonmetals are at the top right. That is, similar elements do not have similar atomic weights.\) The Periodic Table Showing the Elements in Order of Increasing Z This law states that when the elements are listed in order of increasing atomic weights, their properties vary periodically. If a list were made of all elements, we would find the sequence halogen, noble gas, alkali metal, and alkaline-earth metal several more times.ĭmitri Ivanovich Mendeleev proposed the periodic law behind his periodic table compiling. Exactly the same sequence is repeated eight elements later (Ne, Na, and Mg), but this time a halogen (F) precedes the noble gas. The second, third, and forth elements on the list (He, Li, and Be) are a noble gas, an alkali metal, and an alkaline-earth metal, respectively. Obtaining atomic weights, we haveĮlements which belong to families we have already discussed are indicated by shading around their symbols. You can see that such a relationship exist by listing symbols for the first dozen elements in order of increasing relative mass. Therefore it seems reasonable to expect some correlation between this microscopic property and macroscopic chemical behavior. This could account for the related chemical reactivities and analogous compounds of these elements.Īccording to Dalton’s atomic theory, different kinds of atoms may be distinguished by their relative masses (atomic weights). 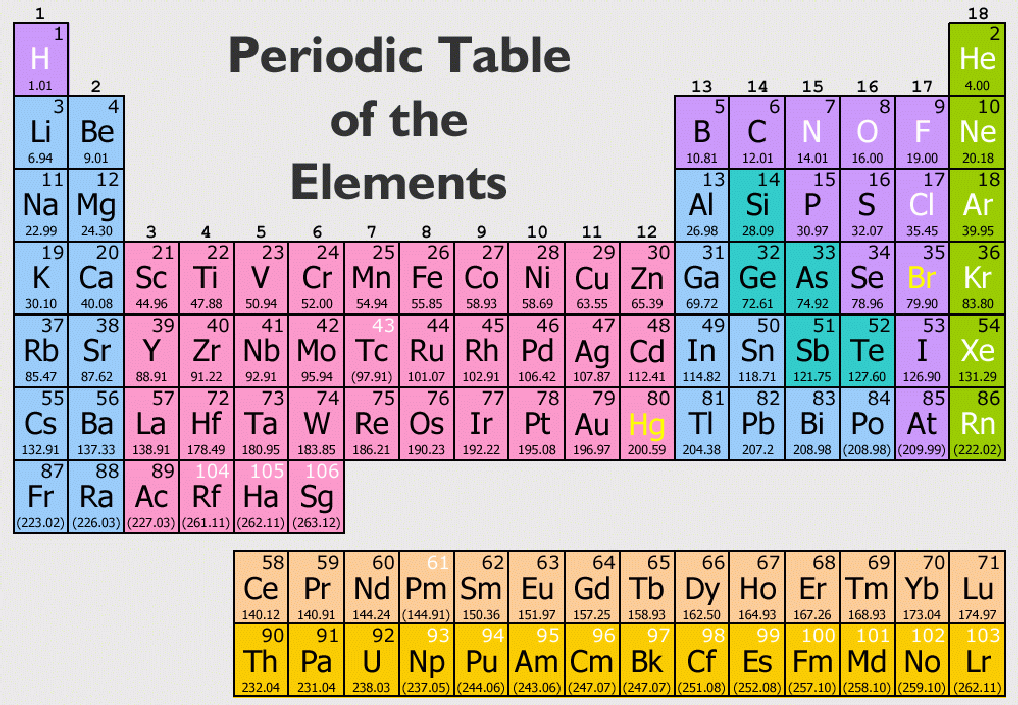 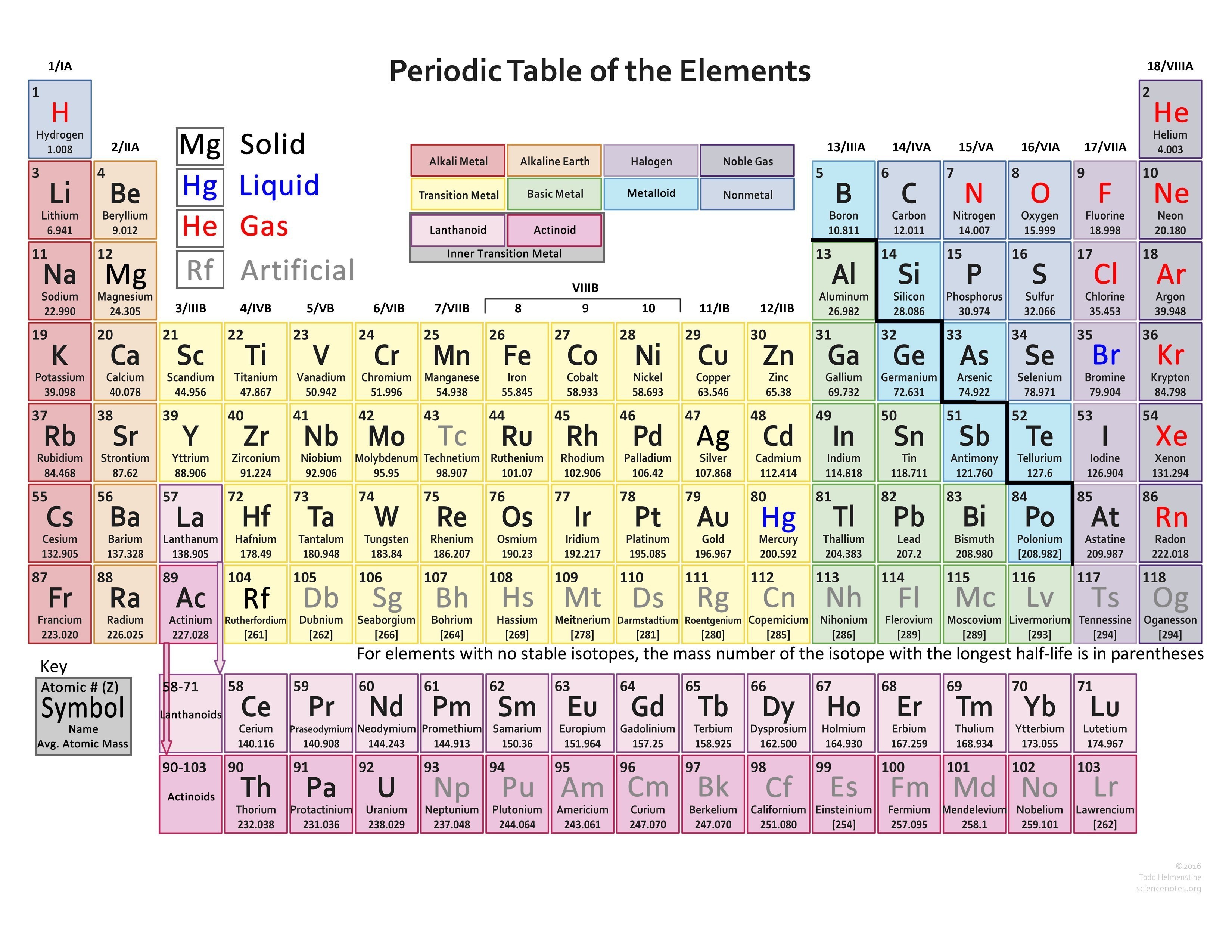
Atoms of sodium ought to be similar in some way to atoms of lithium, potassium, and the other alkali metals. The similarities among macroscopic properties within each of the chemical families lead one to expect microscopic similarities as well. 1950-1999 (+15 elements): Manhattan_Project and Particle physics issues, for atomic numbers 97 and above.1900-1949 (+13 elements): impulse from the old quantum theory, the Refinements to the periodic table, and quantum mechanics.1850-1899 (+23 elements): the age of Classifying Elements received an impulse from the Spectrum analysis.1800-1849 (+22 elements): impulse from Scientific Revolution and Atomic theory and Industrial Revolution.Before 1800 (36 elements): discoveries during and before the Age of Enlightenment.\): Periodic Table showing when each element was discovered 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
